In today’s highly regulated and competitive medical device industry, OEM medical manufacturing has become a strategic solution for brands seeking quality, scalability, and faster market entry.
This is particularly critical in specialized fields such as gynecology and assisted reproductive technology (ART), where precision, safety, and regulatory compliance are essential.
For medical device buyers, distributors, and brand owners, understanding how OEM manufacturing works—and how to select the right partner—is crucial to long-term success.
What Is OEM Medical Manufacturing?
OEM (Original Equipment Manufacturer) medical manufacturing refers to a model in which a specialized manufacturer produces medical devices that are marketed and sold under another company’s brand. While the end customer may recognize the product by a familiar brand name, the design, production, assembly, and quality control are handled by the OEM manufacturer.
In medical devices, OEM manufacturing is far more than simple outsourcing. It requires strict adherence to international quality standards, regulatory frameworks, and clinical performance requirements. Choosing an inexperienced or non-compliant OEM can lead to serious clinical, legal, and reputational risks.
Why OEM Manufacturing Matters in Gynecology and ART
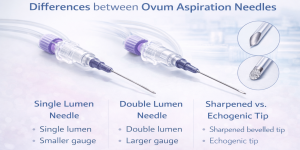
Products such as gynecology catheters, OPU (ovum pick-up) needles, and embryo transfer catheters are used in sensitive clinical procedures where precision and reliability directly affect patient outcomes. For buyers in this field, OEM manufacturing offers several advantages:
- Access to specialized manufacturing expertise
- Reduced capital investment in production facilities
- Faster time to market
- Consistent product quality at scale
However, these benefits are only realized when the OEM partner is experienced, transparent, and fully compliant with medical regulations.
What Buyers Should Look for in an OEM Medical Manufacturer
Choosing an OEM partner is a strategic decision. Buyers should evaluate potential manufacturers based on the following key criteria:
- 1. Regulatory Compliance and Quality Systems
A reliable OEM manufacturer must operate under a certified ISO 13485 quality management system and design products that meet CE and/or FDA requirements, depending on the target market. Proper documentation, traceability, and validation processes are essential. - 2. Product Specialization
Manufacturers with focused expertise in gynecology and ART tend to deliver better clinical performance than general-purpose manufacturers. Experience with OPU needles, embryo transfer catheters, and gynecology devices is a major advantage. - 3. Customization Capabilities
OEM manufacturing often involves adapting products to meet specific customer requirements, including: - Technical specifications
- Packaging design
- Branding and labeling
- Sterilization methods
Flexibility without compromising quality is a key indicator of a strong OEM partner.
- 4. Manufacturing Transparency
Buyers should expect clear communication about materials, processes, testing, and quality controls. Transparency builds trust and ensures smooth, long-term collaboration.
Masstec’s Approach to OEM Medical Manufacturing
Masstec is a specialized medical device manufacturer and brand owner with a strong focus on gynecology and ART devices. We design and produce gynecology catheters, OPU needles, and embryo transfer catheters under the Masstec brand, while also providing OEM medical manufacturing services for international partners.
This dual role provides a unique advantage. As a brand owner, Masstec understands market expectations, regulatory pressures, and clinical demands. As a manufacturer, we deliver controlled production, consistent quality, and scalable output.
Masstec’s OEM manufacturing capabilities include:
- Precision manufacturing of gynecology and ART devices
- Controlled cleanroom environments
- Medical-grade raw materials
- Customizable packaging, labeling, and branding
- Scalable production for global distribution
Unlike manufacturers that focus solely on volume, Masstec prioritizes process control, product validation, and continuous quality improvement. This ensures that OEM partners receive medical devices that meet international standards and perform reliably in clinical settings.
OEM vs. Brand Manufacturing:
Why the Difference Matters
Some buyers assume OEM manufacturing is purely cost-driven. In reality, the most successful OEM partnerships are quality-driven collaborations. Manufacturers that also develop their own branded products, like Masstec, invest more in innovation, regulatory compliance, and long-term reliability.
For distributors and medical brands, working with a company like Masstec means partnering with a manufacturer that is fully accountable for product performance, not just production volume.
The Role of OEM Manufacturing in Global Markets
OEM medical manufacturing plays a vital role in expanding access to high-quality medical devices worldwide. By enabling brands to scale efficiently while maintaining regulatory compliance, OEM partnerships support innovation, affordability, and safety in healthcare systems.
For emerging and established markets alike, OEM manufacturing allows medical brands to focus on clinical education, distribution, and customer support, while relying on expert manufacturers for production excellence.
Final Thoughts
OEM medical manufacturing is not just about producing devices—it is about trust, quality, and shared responsibility. For buyers in gynecology and ART, selecting the right OEM partner directly affects patient safety, clinical outcomes, and brand reputation.
With its dual expertise as a brand owner and OEM manufacturer, Masstec delivers high-quality gynecology catheters, OPU needles, and embryo transfer catheters, ensuring partners receive reliable, compliant, and market-ready medical devices.
Whether you are seeking a trusted OEM partner or branded devices under the Masstec name, our experience, quality systems, and clinical focus make us a preferred choice for international buyers in the reproductive healthcare sector.